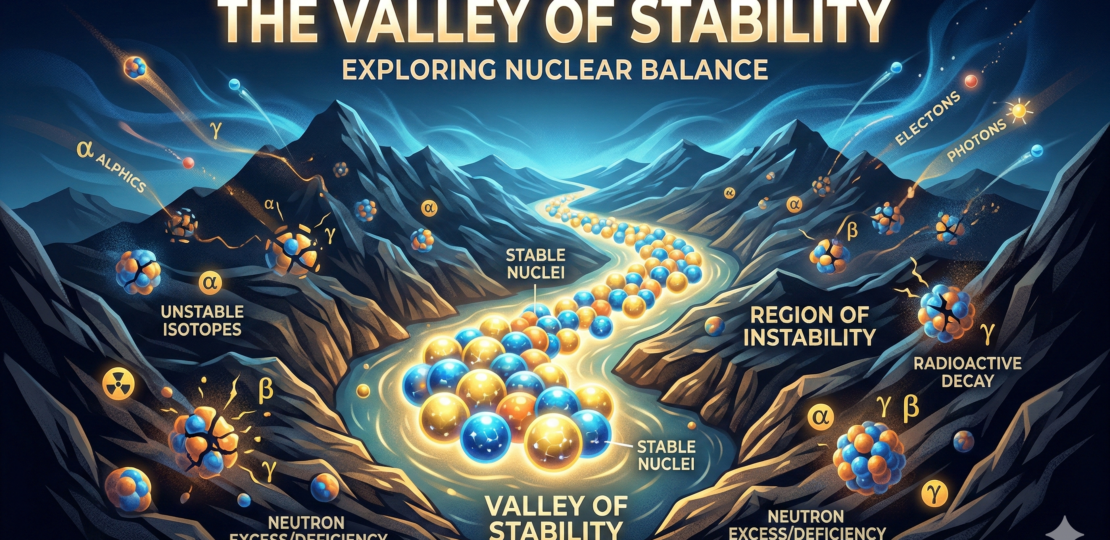
Imagine plotting every possible nucleus on a graph. On one axis: number of protons. On the other: number of neutrons. Each point is a possible combination — some real, some hypothetical, some so unstable they flicker out of existence in less than a blink.
When physicists actually mapped this out, they found something beautiful. The stable nuclei form a curved band across the chart. It’s called the valley of stability.
At low atomic numbers, stability hugs the line where protons equal neutrons. Helium-4? Two and two. Carbon-12? Six and six. Clean symmetry. But as nuclei get heavier, the stable ones start needing more neutrons than protons. Lead-208, for example, has 82 protons but 126 neutrons. That imbalance is not an accident. It’s a survival strategy.
Here’s why.
Protons repel each other electrically. The more protons you pack into a nucleus, the stronger that repulsion becomes. The strong nuclear force holds everything together, but it only acts over extremely short distances. So heavy nuclei need extra neutrons to add more strong-force attraction without adding more electrical repulsion. Neutrons are the silent negotiators in this atomic parliament.
If a nucleus has too many neutrons, it sits above the valley. Too many protons? Below the valley. Nature corrects these imbalances through radioactive decay. Too neutron-heavy? A neutron can convert into a proton via beta decay, emitting an electron. Too proton-heavy? A proton can convert into a neutron, emitting a positron or capturing an electron. The nucleus “slides” back toward stability.
It’s not conscious, obviously. But the metaphor is irresistible. The nucleus seeks balance through transformation.
Push even further from the valley and things get extreme. Some nuclei exist only in particle accelerators. They are born in violent collisions and vanish in milliseconds. These exotic isotopes teach us how nuclear forces behave under stress — like testing a bridge by overloading it. We learn not just where stability lies, but why.
There’s also a delicious irony. Iron-56 sits near the bottom of the valley in terms of binding energy per nucleon. It’s one of the most tightly bound nuclei. Fusion releases energy when you move lighter elements toward iron. Fission releases energy when you split heavier elements toward iron. In a way, iron is the nuclear “sweet spot.” Stars spend their lives climbing toward it. When massive stars finally fill their cores with iron, fusion stops producing energy. Gravity wins. Collapse begins. Supernova.
So this quiet curve on a graph explains both radioactive decay in a lab and the death of stars.
Zoom out and think about it: matter isn’t arbitrary. There’s a landscape of possibility, and stability is a narrow valley running through it. The elements that make up your body — carbon, oxygen, calcium — sit comfortably inside that valley. Just a few neutrons too many or too few, and they would not endure.
Existence, at the nuclear level, is not guaranteed. It is negotiated through forces, probabilities, and balance. And somehow, in that thin valley of stability, complexity gets a foothold and builds everything we know.
RELATED POSTS
View all