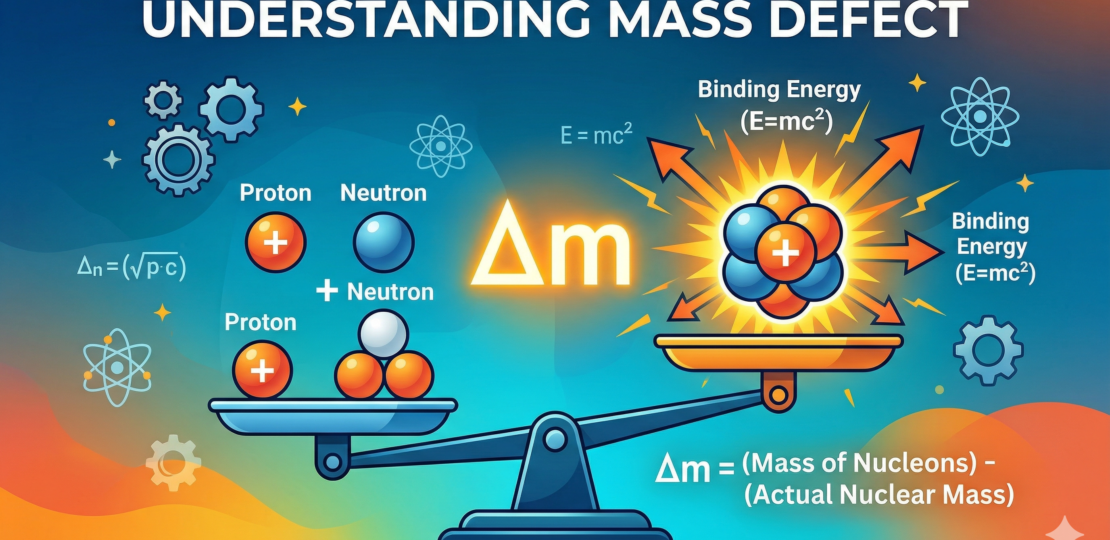
Take a proton and a neutron. Add up their masses. Now bind them together into a nucleus and measure again. You would expect the total to stay the same — after all, nothing has been removed. But the result is surprising: the nucleus weighs slightly less than the sum of its parts. That missing mass is called the mass defect.
This isn’t an error or a mystery gap. The “lost” mass has been converted into energy. This idea comes directly from mass–energy equivalence, which tells us that mass and energy are two forms of the same thing. When nucleons (protons and neutrons) bind together, energy is released, and the system settles into a lower-energy, more stable state. The decrease in mass is simply the energy that has already been given off.
You can think of every stable nucleus as a system that has already “spent” some of its mass to become stable. The stronger the binding, the more energy was released when it formed — and the larger the mass defect. In that sense, mass isn’t just about how much matter is present, but also about how tightly that matter is held together.
This also explains where nuclear energy comes from. In processes like fission or fusion, nuclei rearrange into more stable configurations. The final products have a slightly greater mass defect than the starting ones. That difference in mass is released as energy — sometimes in enormous amounts.
It’s a strange shift in perspective. Mass feels like something fixed and solid, but at the deepest level, it depends on energy. What you hold in your hand is not just matter — it is matter shaped by how much energy has already been transformed and locked into its structure.
RELATED POSTS
View all


