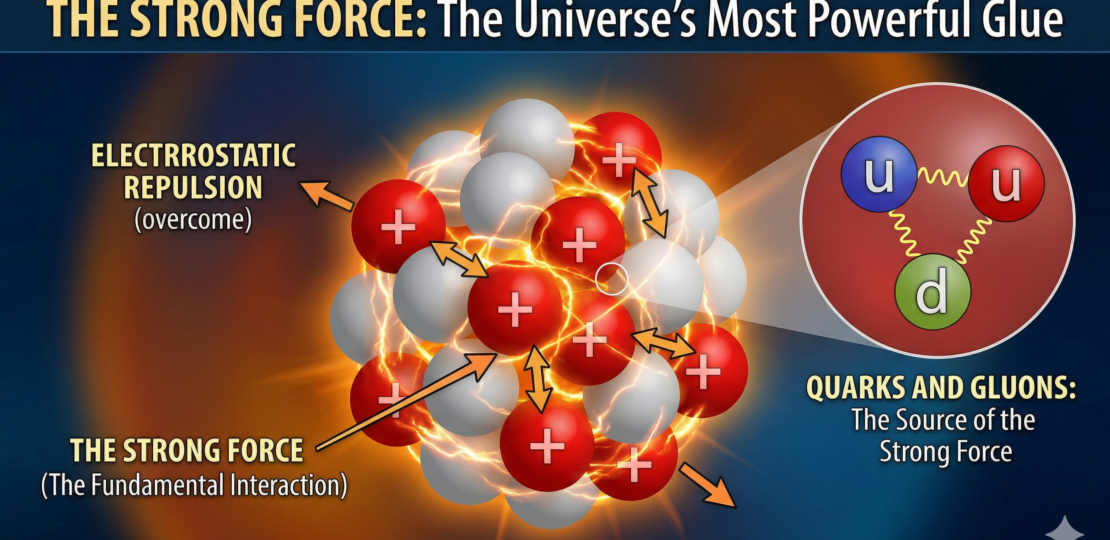
Inside the nucleus, everything depends on a force that is both incredibly powerful and strangely limited. The strong nuclear force is what binds protons and neutrons together, overcoming the intense electrical repulsion between positively charged protons. Without it, no nucleus beyond hydrogen would exist.
But the strong force comes with a catch: it only works over extremely short distances. Think of it as a force that is overwhelming at arm’s length — but disappears if you step even slightly farther away. Unlike gravity or electromagnetism, it doesn’t stretch across space. It’s local, almost intimate.
This creates a natural limit to how large a stable nucleus can be. In small nuclei, every proton and neutron is close enough for the strong force to hold everything tightly together. But as nuclei grow larger, particles on opposite sides become too far apart to feel this force strongly. Meanwhile, electrical repulsion between protons keeps acting across the entire nucleus, pushing everything outward.
So as nuclei get heavier, they face a growing imbalance. The glue weakens with distance, but the repulsion does not. To compensate, nuclei pack in more neutrons, which add attractive strong-force interactions without increasing electrical repulsion. But this trick only works up to a point.
Beyond a certain size, the balance breaks down. The nucleus becomes unstable — prone to decay, fission, or other transformations. This is why elements beyond a certain atomic number are always radioactive. It’s not just bad luck. It’s a structural limitation built into the laws of physics.
There’s something elegant about this constraint. The universe allows complexity, but not without bounds. Even at the smallest scales, there are limits to how much can be held together. Stability isn’t just about strength — it’s about reach.
RELATED POSTS
View all

